CORVIA® ATRIAL SHUNT SYSTEM
A Novel Treatment for Symptomatic Heart Failure
The only direct treatment for high left atrial pressures (LAP)

The Corvia Atrial Shunt is a novel, minimally invasive cardiac implant for patients suffering from Heart Failure with Preserved Ejection Fraction (HFpEF) or Mildy Reduced Ejection Fraction (HFmrEF). Designed to reduce elevated LAP, the primary contributor to HF symptoms, the Corvia Atrial Shunt offers the most advanced therapeutic option for HF patients with an EF≥40%.
The Corvia Atrial Shunt is the most widely studied interatrial shunt for heart failure. It has been implanted in over 675 patients worldwide, and more than 250 patients have had the shunt for over 5 years.1
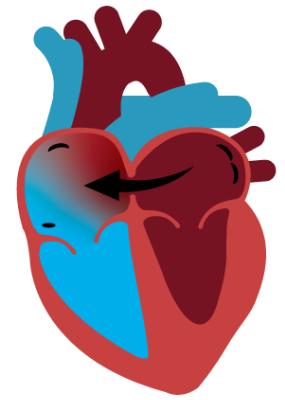
Dynamic Decompression
Atrial shunting safely relieves the high pressures that cause HF symptoms by connecting the two upper chambers of the heart. This connection allows the heart to dynamically decompress by directing blood from the left to right atrium on demand, thereby reducing HF symptoms.
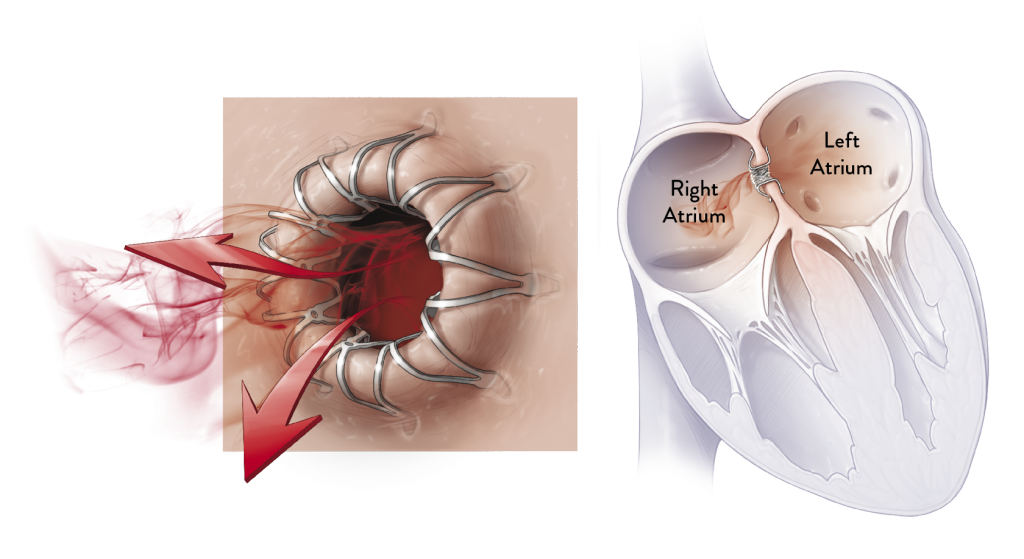
HOW IT WORKS
The Corvia Atrial Shunt is placed by an interventional
cardiologist or electrophysiologist during a one-time
procedure. A catheter is used to place a Corvia Atrial Shunt
as a passage between the left and right atria. This passage
allows blood to flow from the high pressure left atrium to the
lower pressure right atrium, thereby reducing pressure in the
lungs and left side of the heart.
WATCH HOW IT WORKS
By facilitating continuous and dynamic decompression of the left atrium, the Corvia Atrial Shunt is intended to reduce heart failure hospitalizations and improve heart failure symptoms and quality of life.2
IMPORTANT SAFETY INFORMATION
CLICK HERE for the Instructions for Use and Magnetic Resonance (MR) Imaging information.
- Unpublished data as of March 1, 2025 compiled from Corvia Clinical Trials and on file at Corvia Medical.
- Gustafsson, F, Petrie, M, Komtebedde, J. et al. 2-Year Outcomes of an Atrial Shunt Device in HFpEF/HFmrEF: Results From REDUCE LAP-HF II. J Am Coll Cardiol HF. 1 Jun 2024. https://doi.org/10.1016/j.jchf.2024.04.011
